Acorda Therapeutics Overview
The Overall Ranking is a score from 1 star (very bad) to 5 stars (excellent) generated based on the Company Reviews of current and former employees at this company, taking everything into account.
The number you see in the middle of the donut pie chart is the simple average of these scores. If you hover over the various sections of the donut, you will see the % breakdown of each score given.
The percentile score in the title is calculated across the entire Company Database and uses an adjusted score based on Bayesian Estimates (to account for companies that have few reviews). Simply put, as a company gets more reviews, the confidence of a "true score" increases so it is pulled closer to its simple average and away from the average of the entire dataset.
- 5 Stars
- 4 Stars
- 3 Stars
- 2 Stars
- 1 Star
Acorda Therapeutics Job Openings
No jobs found.
Company Details
Acorda Therapeutics is a biotechnology company focused on developing therapies that restore function and improve the lives of people with MS, spinal cord injury and other neurological conditions.
Acorda markets AMPYRA® (dalfampridine) Extended Release Tablets, 10 mg, in the United States as a treatment to improve walking in patients with multiple sclerosis (MS). This was demonstrated by an improvement in walking speed. AMPYRA is marketed outside the United States as FAMPYRA® (prolonged-release fampridine tablets) by Biogen Idec under a licensing agreement from Acorda. AMPYRA and FAMPYRA are manufactured under license from Alkermes Pharma Ireland Limited.
The Company also markets ZANAFLEX CAPSULES® (tizanidine hydrochloride) and Zanaflex tablets, a short-acting drug for the management of spasticity. Acorda also receives sales royalties on tizanidine hydrochloride capsules, an authorized generic version of ZANAFLEX CAPSULES distributed by Watson Pharmaceutics, Inc. under its agreement with Acorda.
Acorda has an industry-leading pipeline of novel neurological therapies. The Company is developing Diazepam Nasal Spray for treatment of certain epileptic seizures. It is also studying AMPYRA to improve a range of functional impairments caused by MS, as well as its potential for use in other neurological conditions, including cerebral palsy and post-stroke deficits. In addition, Acorda is developing clinical stage compounds AC105 for acute treatment of spinal cord injury, GGF2 for treatment of heart failure and rHIgM22, a remyelinating monoclonal antibody, for the treatment of MS. GGF2 is also being investigated in preclinical studies as a treatment for neurological conditions such as stroke and spinal cord injury. Chondroitinase, an enzyme that encourages nerve plasticity in spinal cord injury, is in preclinical development.
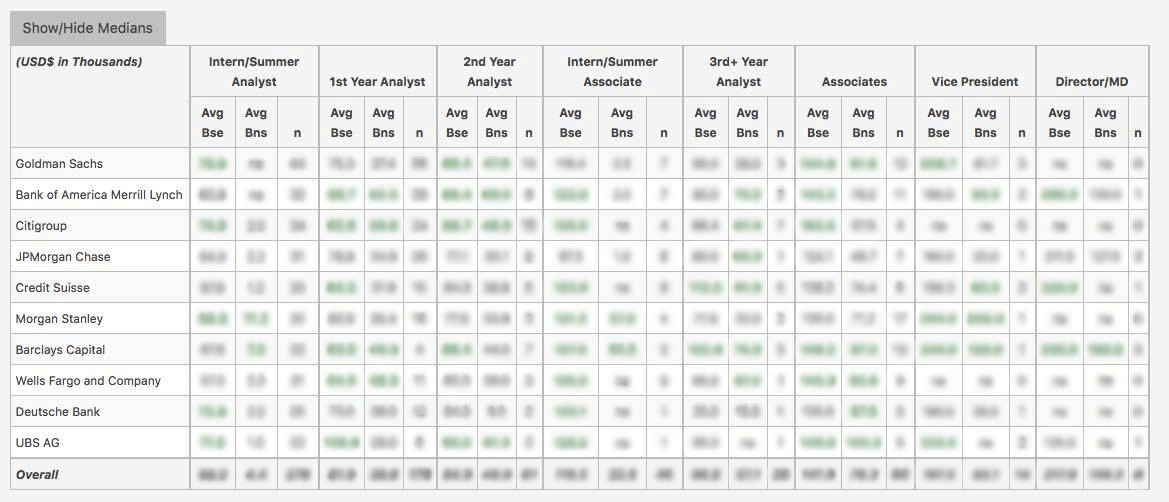
WSO Company Database Comparison Table
Acorda Therapeutics Interview Questions




or Want to Sign up with your social account?